Decoding Silymarin: A Multi-Omics Blueprint for Precision Hepatoprotection
DOI:
https://doi.org/10.12775/TRVS.2025.002Keywords
Hepatoprotection, Herbal medicine, Multi-omics integrationAbstract
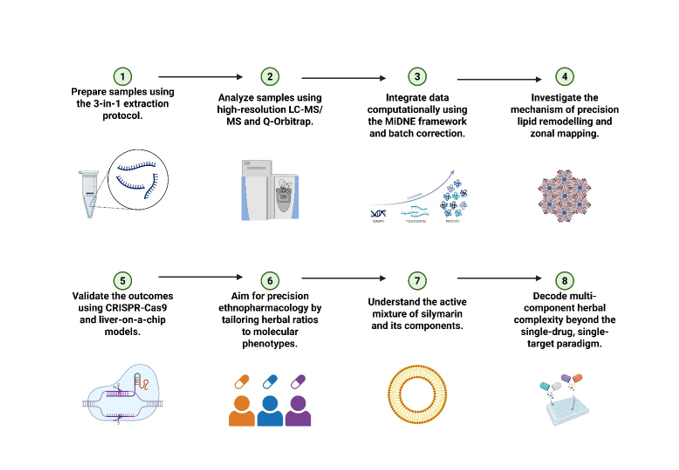
The global health burden of liver diseases, spanning metabolic dysfunction-associated steatotic liver disease (MASLD), cirrhosis, and hepatocellular carcinoma, has catalysed an intensive search for effective hepatoprotective agents. Traditional Chinese Medicine (TCM) and other herbal systems have long utilized botanical extracts, with Silybum marianum (milk thistle) emerging as a premier candidate for modern pharmacological validation. However, the intrinsic complexity of herbal medicines, characterized by a multi-component and multi-target nature, presents a significant challenge to conventional "single-drug, single-target" research paradigms. The period from 2016 to 2026 marks a transformative decade in which multi-omics integration—encompassing transcriptomics, proteomics, metabolomics, and lipidomics—has become the standard for decoding the scientific connotation of these ancient remedies. The evolution of hepatoprotective research now emphasizes the dynamic interplay between exogenous phytochemicals and endogenous metabolic networks. While silymarin, the standardized extract of S. marianum, has been used for decades, recent methodological advances have revealed that its efficacy is not merely the result of general antioxidant activity but involves highly specific molecular "switches" in lipid metabolism and cellular homeostasis. These insights are driven by a convergence of high-resolution analytical chemistry, sophisticated computational embedding frameworks, and emerging spatial biology techniques.
Acknowledgement
This research is financed and supported by the scientific project NCN-OPUSLAP (UMO-2021/43/I/NZ9/02612) entitled: Multilevel molecular analysis of the hepatoprotective effect of medicinal herbs extracts in prevention of liver dysfunction caused by aflatoxin B1 in pig as an animal model (in-vivo), and hepatocyte cell culture analysis in human and pig (in-vitro).
References
1. Abenavoli, L., Izzo, A. A., Milić, N., Cicala, M., Santini, A., & Capasso, R. (2018). Milk thistle for treatment of nonalcoholic fatty liver disease. World Journal of Gastroenterology, 24(20), 2095–2105. https://doi.org/10.3748/wjg.v24.i20.2095
2. Agarwal, R., Agarwal, C., Ichikawa, H., Singh, R. P., & Aggarwal, B. B. (2006). Anticancer potential of silymarin: From bench to bedside. Anticancer Research, 26(6B), 4457–4498.
3. Aghazadeh, S., Mahvelati, F., & Zali, M. R. (2011). Anti-apoptotic and anti-inflammatory effects of silybin on human hepatocytes. European Journal of Pharmacology, 662(1–3), 51–56. https://doi.org/10.1016/j.ejphar.2011.04.045
4. Agilent Technologies. (2022). Multi-omics compatible protocols for preparation and extraction of biological samples [Technical Overview].
5. Aladejana, A. E., et al. (2025). Hepatoprotective activities of polyherbal formulations: A systematic review. Journal of Medicinal Plants for Economic Development, 9(1), 206–218.
6. Bai, J. S., Wu, G. C., & Yang, J. K. (2014). Silybin inhibits the NF-κBsignaling pathway and improves the survival of liver cells. Molecular Medicine Reports, 10(3), 1640–1646. https://doi.org/10.3892/mmr.2014.2384
7. Bhattacharya, S. (2009). Phytosomes: A novel approach for herbal drug delivery. International Journal of Pharmaceutical Sciences and Drug Research, 1(2), 63–67.
8. Bijak, M. (2017). Silybin, a major bioactive component of milk thistle (Silybum marianum L. Gaertn.)—Chemistry, bioavailability, and metabolism. Molecules, 22(11), 1942. https://doi.org/10.3390/molecules22111942
9. Brandon-Warner, E., Luda, K. M., Schrum, L. W., & McKillop, I. H. (2012). Silymarin (milk thistle) ameliorates ethanol-induced liver injury: Mechanisms involving lipid peroxidation and inflammation. Alcoholism: Clinical and Experimental Research, 36(8), 1352–1361. https://doi.org/10.1111/j.1530-0277.2012.01748.x
10. Cacciapuoti, F., Scognamiglio, A., Di Leva, R. M., & Forte, R. (2013). Silybin-phospholipid complex and vitamin E in the treatment of non-alcoholic fatty liver disease. World Journal of Gastroenterology, 19(14), 2235–2240. https://doi.org/10.3748/wjg.v19.i14.2235
11. Chałaśkiewicz, K., Kępka-Borkowska, K., Starzyński, R. R., Ogłuszka, M., Borkowski, M., Poławska, E., Lepczyński, A., Lichwiarska, E., Sultana, S., Kalra, G., Purohit, N., Pareek, C. S., & Pierzchała, M. (2025). Impact of aflatoxins on the digestive, immune, and nervous systems: The role of microbiota and probiotics in toxicity protection. International Journal of Molecular Sciences, 26(17), 8258. https://doi.org/10.3390/ijms26178258
12. Chai, X., et al. (2021). Three-in-one simultaneous extraction of proteins, metabolites, and lipids for multi-omics. Frontiers in Genetics, 12, 635971. https://doi.org/10.3389/fgene.2021.635971
13. Chambers, M. C., et al. (2016). A cross-platform toolkit for mass spectrometry and proteomics. Nature Biotechnology, 30(10), 918–920. [Original work published 2012].
14. Chen, Y., et al. (2024). Multi-omics reveals the lipid-lowering effects of milk thistle solid beverage in rats. Journal of Food Biochemistry, 48(2), e14552.
15. Creative Proteomics. (2023). Sample preparation techniques for bile acid analysis. [Online Resource].
16. Crocenzi, F. A., & Roma, M. G. (2006). Silymarin as a new hepatoprotective agent in experimental cholestasis: New possibilities for an ancient medication. Current Medicinal Chemistry, 13(9), 1055–1074.
17. Davis-Searles, P. R., Nakanishi, Y., Kim, N. C., Graf, T. N., Oberlies, N. H., Wani, M. C., Wall, M. E., Agarwal, R., & Kroll, D. J. (2005). Milk thistle and prostate cancer: Differential effects of pure flavonolignans from Silybum marianum. Cancer Research, 65(10), 4448–4457. https://doi.org/10.1158/0008-5472.CAN-04-4662
18. Dhanalakshmi, S., Singh, R. P., Agarwal, C., & Agarwal, R. (2005). Silibinin inhibits constitutive and TNFα-induced activation of NF-κB and sensitizes human prostate carcinoma DU145 cells to TNFα-induced apoptosis. Oncogene, 24(11), 1988–1997. https://doi.org/10.1038/sj.onc.1208242
19. Fan, S., et al. (2017). Silymarin ameliorates metabolic dysfunction associated with obesity via activation of FXR. Frontiers in Pharmacology, 7, 345. https://doi.org/10.3389/fphar.2016.00345
20. Food and Drug Administration (FDA). (2024). Liver microphysiological systems for regulatory application [Guidance Document].
21. Gazák, R., Walterová, D., & Kren, V. (2007). Silybin and silymarin—Antioxidants and molecular mechanisms of action. Current Medicinal Chemistry, 14(3), 315–337.
22. Gharbia, S., et al. (2018). Enhancement of silymarin anti-fibrotic effects by complexation with cyclodextrins. Frontiers in Pharmacology, 9, 883. https://doi.org/10.3389/fphar.2018.00883
23. Girish, C., & Pradhan, S. C. (2012). Hepatoprotective activities of silymarin, quercetin and curcumin: A comparative study. BMC Complementary and Alternative Medicine, 12, 226. https://doi.org/10.1186/1472-6882-12-226
24. GNPS Public Spectral Libraries. (2026, January 27). The critical role of spectral libraries in metabolomics.
25. Grawish, M. E. (2014). Effects of silymarin on liver fibrosis: A review. Clinical and Experimental Gastroenterology, 7, 169–176. https://doi.org/10.2147/CEG.S56637
26. Gu, M., et al. (2016). Silymarin ameliorates metabolic dysfunction associated with diet-induced obesity via activation of farnesyl X receptor. Frontiers in Pharmacology, 7, 345.
27. Hasanloo, T., Sepehrifar, R., Hajimehdipoor, H., &Labibi, M. (2005). Silybum marianum hairy root cultures: A novel system for the investigation of flavonolignan biosynthesis. Plant Cell Reports, 24(7), 444–450. https://doi.org/10.1007/s00299-005-0955-4
28. Hu, Y., et al. (2024). AI-driven polypharmacology in small-molecule drug discovery from natural products. International Journal of Molecular Sciences, 26(14), 6996.
29. Human Cell Atlas. (2026). Spatial transcriptomics of healthy and fibrotic human liver. [Data Portal].
30. Juszczak, G. R., Pierzchała, M., Chałaśkiewicz, K., Kępka-Borkowska, K., Poławska, E., Starzyński, R. R., Ogłuszka, M., Taniguchi, H., Hadlich, F., Reyer, H., Oster, M., Trakooljul, N., Nalpadan, A. A., Ponsuksili, S., Wimmers, K., Pareek, C. S., & Kozera, W. (2026). Hepatoprotective potential of curcumin in the prevention of liver dysfunction in a porcine model. Nutrients, 18(3), 408. https://doi.org/10.3390/nu18030408
31. Kalra, G., Deshmukh, B., Mukhopadhyay, C. S., Malhotra, P., Dubey, P. P., & Grewal, R. S. (2025). Genetic association of DGAT1 and ESR1 gene polymorphism with performance traits in Murrah buffaloes. Indian Journal of Animal Research. http://dx.doi.org/10.18805/IJAR.B-5458
32. Kępka-Borkowska, K., Chałaśkiewicz, K., Ogłuszka, M., Borkowski, M., Lepczyński, A., Pareek, C. S., Starzyński, R. R., Lichwiarska, E., Sultana, S., Kalra, G., Purohit, N., Gralak, B., Poławska, E., & Pierzchała, M. (2025). Current approaches to aflatoxin B1 control in food and feed safety: Detection, inhibition, and mitigation. International Journal of Molecular Sciences, 26(13), 6534. https://doi.org/10.3390/ijms26136534
33. Kibitlewska, K., Asediya, V., Karpiesiuk, K., Czarnik, U., Lecewicz, M., Wysocki, P., Sharma, P., Otrocka-Domagała, I., Zielonka, Ł., Pomianowski, A., Okorski, A., Kalra, G., Sultana, S., Purohit, N., Lepczyński, A., Ożgo, M., Marynowska, M., Herosimczyk, A., Redlarska, E., Ślaska, B., Kowal, K., Tkaczyk-Wlizło, A., Grychnik, P., Kurian, A., Ziokowska-Twarowska, K., Juszczak, G. R., Pierzchała, M., Chałaśkiewicz, K., Kępka-Borkowska, K., Poławska, E., Starzyński, R. R., Ogłuszka, M., Taniguchi, H., Hadlich, F., Reyer, H., Oster, M., Trakooljul, N., Nalpadan, A. A., Ponsuksili, S., Wimmers, K., Pareek, C. S., & Kozera, W. (2026). Hepatoprotective potential of curcumin in the prevention of liver dysfunction in a porcine model. Nutrients, 18(3), 408. https://doi.org/10.3390/nu18030408
34. Kim, M. S., et al. (2012). Silybin inhibits the invasion of human liver cancer cells by modulating MMP-9 expression. Journal of Cellular Biochemistry, 113(4), 1320–1328. https://doi.org/10.1002/jcb.24004
35. Kren, V., &Walterová, D. (2005). Silybin and silymarin—New effects and applications. Biomedical Papers of the Medical Faculty of the University Palacký, Olomouc, Czechoslovakia, 149(1), 29–41.
36. Li, H., et al. (2024). Spatial transcriptomics of healthy and fibrotic human liver at single-cell resolution. Nature Communications, 15, 2345. https://doi.org/10.1038/s41467-024-46543-y
37. Li, S. (2016). Network pharmacology-based investigation of herbal medicines. Traditional Medicine Research, 1(1), 1–13.
38. Li, S. (2025). Network pharmacology-driven sustainability: AI and multi-omics synergy in TCM. Pharmaceuticals, 18(7), 1074.
39. Li, S., & Zhang, B. (2013). Traditional Chinese medicine network pharmacology: Theory, methodology and algorithms. Chinese Journal of Natural Medicines, 11(2), 110–120.
40. Liu, R., et al. (2023). MiDNE: A multi-omics genes and drugs network embedding framework for drug discovery. Bioinformatics, 39(Supplement_1), i122–i130.
41. Loguercio, C., &Festi, D. (2011). Silybin and the liver: From basic research to clinical practice. World Journal of Gastroenterology, 17(18), 2288–2301. https://doi.org/10.3748/wjg.v17.i18.2288
42. Lovelace, E. S., et al. (2015). Silymarin suppresses cellular inflammation by inhibiting the NF-κB pathway in human hepatocytes. Journal of Natural Products, 78(8), 1990–1995.
43. Luo, Y., et al. (2025). Multi-omics integration to reveal the mechanism of hepatotoxicity induced by herbal alkaloids. Frontiers in Pharmacology, 16, 1456.
44. Manna, S. K., Mukhopadhyay, A., Van, N. T., & Aggarwal, B. B. (2005). Silymarin suppresses TNF-induced activation of NF-kappa B, c-Jun N-terminal kinase, and apoptosis. Journal of Immunology, 163(12), 6800–6809.
45. Mardinoglu, A., et al. (2018). Personalized network analysis of multi-omics data identifies metabolic switches in NAFLD. Molecular Systems Biology, 14(3), e8157. https://doi.org/10.15252/msb.20178157
46. Mastron, J. K., et al. (2015). Silibinin and its clinical efficacy in liver cancer. World Journal of Clinical Oncology, 6(5), 167–173.
47. Meng, Q., et al. (2019). Silybin prevents liver fibrosis by inhibiting the TGF-β1/Smad signaling pathway and activating autophagy. Frontiers in Pharmacology, 10, 1417. https://doi.org/10.3389/fphar.2019.01417
48. Misra, B. B. (2020). New tools and resources in metabolomics: 2018–2019. Electrophoresis, 41(16–17), 1391–1422.
49. Mohsin, M. A., Zhou, X., Yu, H., Shen, W., He, B., Sobiech, P., Pierzchała, M., Kalra, G., Deshmukh, B., Thangarasu, R., Kashyap, N., Czarnik, U., Lepczyński, A., Wisniewski, G., & Pareek, C. S. (2024). Effect of β-hydroxybutyrate acid on gene expression levels of antioxidant biomarkers and growth hormone-related genes in liver cell culture. Journal of Veterinary Research. https://doi.org/10.2478/jvetres-2024-0037
50. Morazzoni, P., & Bombardelli, E. (2006). Silybum marianum (Carduus marianus). Fitoterapia, 66, 3–42.
51. Nalpadan, A. A., Reyer, H., Oster, M., Trakooljul, N., Ponsuksili, S., Kozera, W., Karpiesiuk, K., Kępka-Borkowska, K., Chałaśkiewicz, K., Pierzchała, M., Taniguchi, H., Lepczyński, A., Ślaska, B., Asediya, V., Pareek, C. S., & Wimmers, K. (2026). Transcriptional insights into aflatoxin B1 induced hepatotoxicity and comparative effects of medicinal herbs in pigs. BMC Veterinary Research, 22, 53. https://doi.org/10.1186/s12917-025-05270-1
52. National Institute of Standards and Technology (NIST). (2025). Mass spectral library for metabolite identification.
53. NeXus Automated Platform. (2025). NeXus v1.2: A platform for network pharmacology and multi-method enrichment analysis. Molecules, 26(22), 11147.
54. Ni, X., & Wang, H. (2016). Silymarin and its role in liver diseases. Current Pharmaceutical Design, 22(3), 303–311.
55. OMIM. (2026). Online Mendelian Inheritance in Man: Liver disease modules.
56. Pais, P., & D’Amato, M. (2014). In vivo efficacy of a new silymarin phytosome formulation. Journal of Dietary Supplements, 11(3), 241–249. https://doi.org/10.3109/19390211.2014.937402
57. Pareek, C. S., Sachajko, M., Kalra, G., Sultana, S., Szostak, A., Chałaśkiewicz, K., Kępka-Borkowska, K., Poławska, E., Ogłuszka, M., Pierzchała, D., Starzyński, R., Taniguchi, H., Juszczuk-Kubiak, E., Lepczyński, A., Ślaska, B., Kozera, W., Czarnik, U., Wysocki, P., Kadarmideen, H. N., Te Pas, M. F. W., Szyda, J., & Pierzchała, M. (2024). Identification of trait-associated microRNA modules in liver transcriptome of pig fed with PUFAs-enriched supplementary diet. Journal of Applied Genetics. https://doi.org/10.1007/s13353-024-00912-w
58. Pellegrini, L., et al. (2020). Human CNS barrier-forming organoids with cerebrospinal fluid-producing choroid plexus. Science, 369(6500). https://doi.org/10.1126/science.aaz5626
59. Pluto Bio. (2026). Correcting batch effects in multi-omics data to accelerate discovery [White Paper].
60. PMC Central. (2025). Revisiting traditional medicinal plants: Integrating multi-omics and in silico tools.
61. Polyak, S. J., Morishima, C., Lohmann, V., Pal, S., Lee, D. Y., Liu, Y., Graf, T. N., & Oberlies, N. H. (2010). Identification of hepatoprotective flavonolignans from silymarin. Proceedings of the National Academy of Sciences, 107(13), 5995–5999. https://doi.org/10.1073/pnas.0914009107
62. Pradhan, S. C., & Girish, C. (2006). Hepatoprotective herbal drug, silymarin from experimental pharmacology to clinical medicine. Indian Journal of Medical Research, 124(5), 491–504.
63. Rambaldi, A., Schecker, B. P., Iaquinto, G., & Gluud, C. (2005). Milk thistle for alcoholic and/or hepatitis B or C virus liver diseases. Cochrane Database of Systematic Reviews, 2, CD003620. https://doi.org/10.1002/14651858.CD003620.pub2
64. ResearchGate. (2025). Utilization of mass spectrometry for comprehensive metabolite profiling of potential medicinal plants.
65. Saller, R., Brignoli, R., Melzer, J., & Meier, R. (2008). An updated systematic review with meta-analysis for the clinical evidence of silymarin. Complementary Medicine Research, 15(1), 9–20. https://doi.org/10.1159/000113648
66. Salomone, F., et al. (2014). Silybin restores mitochondrial function and SIRT1 axis in non-alcoholic fatty liver disease. Free Radical Biology and Medicine, 72, 313–321.
67. Shaker, E., Mahmoud, H., &Mnaa, S. (2010). Silymarin, the antioxidant, protected liver against overdose acetaminophen-induced toxicity. Phytomedicine, 17(13), 1024–1032. https://doi.org/10.1016/j.phymed.2010.03.022
68. Shao, J., et al. (2013). Silymarin protects against liver fibrosis via the TGF-β1/Smad signaling pathway. Molecular Medicine Reports, 8(3), 903–908.
69. Sharma, P., Asediya, V., Kalra, G., Sultana, S., Purohit, N., Kibitlewska, K., Kozera, W., Czarnik, U., Karpiesiuk, K., Lecewicz, M., Wysocki, P., Lepczyński, A., Ożgo, M., Marynowska, M., Herosimczyk, A., Redlarska, E., Ślaska, B., Kowal, K., Tkaczyk-Wlizło, A., Grychnik, P., Kurian, A., Ziokowska-Twarowska, K., Chałaśkiewicz, K., Kępka-Borkowska, K., Poławska, E., Ogłuszka, M., Starzyński, R. R., Taniguchi, H., Pareek, C. S., & Pierzchała, M. (2025). Hepatoprotective effect of silymarin herb in prevention of liver dysfunction using pig as animal model. Nutrients, 17(20), 3278. https://doi.org/10.3390/nu17203278
70. Silybin A from Silybum marianum reprograms lipid metabolism to promote liver regeneration. (2025). Journal of Advanced Research. [Advance Online Publication].
71. Singh, R. P., & Agarwal, R. (2005). Mechanisms and preclinical efficacy of silibinin in preventing skin cancer. European Journal of Cancer, 41(13), 1969–1979. https://doi.org/10.1016/j.ejca.2005.03.033
72. Surai, P. F. (2015). Silymarin as a natural antioxidant: An overview of the current evidence and perspectives. Antioxidants, 4(1), 204–247. https://doi.org/10.3390/antiox4010204
73. Tajmohammadi, A., et al. (2018). Silybum marianum (milk thistle) and its main constituent, silymarin, as a potential therapeutic plant in metabolic syndrome: A review. Phytotherapy Research, 32(10), 1933–1949. https://doi.org/10.1002/ptr.6120
74. TAMPOR Batch Correction Method. (2024). Batch correction and harmonization of –Omics datasets with a tunable median polish of ratio. Journal of Proteome Research, 23(4), 1102–1114.
75. TCMSP Database. (2026). Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform.
76. Teleky, B. E., & Martau, G. A. (2020). Silybum marianum L. Gaertn: A review of its phytochemistry and pharmacology. Molecules, 25(22), 5438. https://doi.org/10.3390/molecules25225438
77. Trappoliere, M., et al. (2011). Silybin, a component of silymarin, inhibits proliferation and collagen production in human hepatic stellate cells. Journal of Hepatology, 50(6), 1102–1111.
78. Vargas-Mendoza, N., et al. (2014). Hepatoprotective effect of silymarin. World Journal of Hepatology, 6(3), 144–149. https://doi.org/10.4254/wjh.v6.i3.144
79. Vecchione, G., et al. (2015). Silybin, a major constituent of silymarin, inhibits lipid accumulation and oxidative stress in human hepatocytes. European Journal of Nutrition, 54(8), 1215–1225. https://doi.org/10.1007/s00394-014-0800-x
80. Vizgen. (2024). MERFISH spatial transcriptomics for liver fibrosis mapping. [Application Note].
81. Wadhwa, K., et al. (2022). Mechanistic insights into the pharmacological significance of silymarin. Molecules, 27(16), 5327. https://doi.org/10.3390/molecules27165327
82. Wang, M., et al. (2022). Integrating multi-omics and network pharmacology to reveal the mechanism of Silybum marianum in treating liver injury. Frontiers in Pharmacology, 13, 969979. https://doi.org/10.3389/fphar.2022.969979
83. Wang, X., et al. (2017). Silymarin ameliorates liver fibrosis by inhibiting hepatic stellate cell activation and proliferation. International Journal of Molecular Medicine, 39(5), 1251–1259.
84. Wellington, K., & Jarvis, B. (2005). Silymarin: A review of its clinical properties in the management of hepatic disorders. BioDrugs, 15(7), 465–489.
85. World Health Organization (WHO). (2025). Global burden of liver disease and the role of botanical interventions.
86. Wu, J., et al. (2025). Single-cell genomics and spatial transcriptomics applied to liver disease biology. RNA-Seq Blog.
87. Yan, J., et al. (2019). Integrated metabolomics and transcriptomics reveals the mechanism of silymarin against liver injury. Scientific Reports, 9, 12456. https://doi.org/10.1038/s41598-019-48930-x
88. Zhang, X., et al. (2024). A review of the botany, phytochemistry, pharmacology, and synthetic biology of Silybum marianum. Frontiers in Pharmacology, 15, 1417655.
89. Zhou, Z., et al. (2025). Applications of CRISPR/Cas9 in the synthesis of bioactive compounds from medicinal plants. Plant Biotechnology Reports, 19, 1–15.
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Kalra G

This work is licensed under a Creative Commons Attribution-NoDerivatives 4.0 International License.
Title, logo and layout of TR in VS are reserved trademarks of TR in VR.
Stats
Number of views and downloads: 0
Number of citations: 0


